 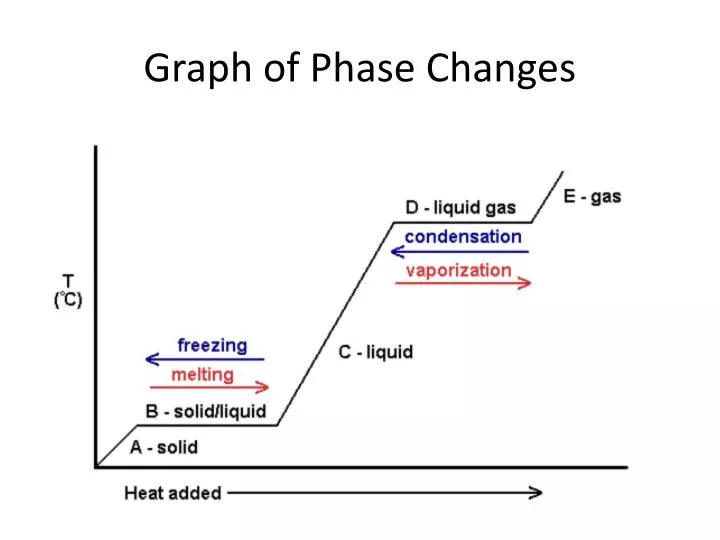
Phase transitions occur when the thermodynamic free energy of a system is non-analytic for some choice of thermodynamic variables (cf. When water vapor condenses (an equilibrium fractionation), the heavier water isotopes ( 18O and 2H) become enriched in the liquid phase while the lighter isotopes ( 16O and 1H) tend toward the vapor phase. Isotope fractionation occurs during a phase transition, the ratio of light to heavy isotopes in the involved molecules changes. 
The breaking of symmetries in the laws of physics during the early history of the universe as its temperature cooled.The superfluid transition in liquid helium is an example of this. Quantum condensation of bosonic fluids ( Bose–Einstein condensation).The transition between different molecular structures ( polymorphs, allotropes or polyamorphs), especially of solids, such as between an amorphous structure and a crystal structure, between two different crystal structures, or between two amorphous structures.The emergence of superconductivity in certain metals and ceramics when cooled below a critical temperature.The dependence of the adsorption geometry on coverage and temperature, such as for hydrogen on iron (110).Order-disorder transitions such as in alpha- titanium aluminides.Changes in the crystallographic structure such as between ferrite and austenite of iron.The martensitic transformation which occurs as one of the many phase transformations in carbon steel and stands as a model for displacive phase transformations.The transition between differently ordered, commensurate or incommensurate, magnetic structures, such as in cerium antimonide.The transition between the ferromagnetic and paramagnetic phases of magnetic materials at the Curie point.Transition to a mesophase between solid and liquid, such as one of the " liquid crystal" phases.A spinodal decomposition, in which a single phase is cooled and separates into two different compositions of that same phase.A peritectic transformation, in which a two-component single-phase solid is heated and transforms into a solid phase and a liquid phase.A metastable polymorph which forms rapidly due to lower surface energy will transform to an equilibrium phase given sufficient thermal input to overcome an energetic barrier. A metastable to equilibrium phase transformation.The same process, but beginning with a solid instead of a liquid is called a eutectoid transformation. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
